Duration
11:00 minutes
Category
Stem cell basics
Series
Cord blood banking
Objective
Educational
What you will learn
- What is cord blood banking?
- What are stem cells?
- Clinical trial involving newborn stem cells
- Introduction to stem cell therapy
- 3 easy steps to cord blood banking
Understanding cord blood and cord tissue banking
This webinar will focus on cord blood and cord tissue banking to give you an understanding of the process and its importance.
Here is a quick overview of today’s seminar. We will start by highlighting the difference between cord blood and cord tissue, then introduce you to stem cells more specifically newborn stem cells. Discuss how stem cells are currently being used in the clinic to treat diseases and how scientists are exploring their potential for future applications in the lab. We will end by a quick recap of process involved in cord blood and cord tissue banking.
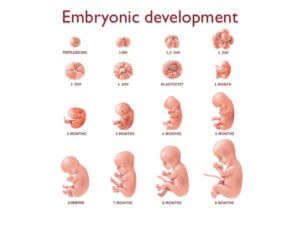 We are going to begin with a glimpse into embryonic development. Once conceived, a baby goes through several different stages of development; every baby starts off as a single fertilized egg cell.
We are going to begin with a glimpse into embryonic development. Once conceived, a baby goes through several different stages of development; every baby starts off as a single fertilized egg cell.
Then within the span of a few of weeks the embryo goes on to establish the basic body plan of an adult. Once this is done, it is a matter of growing and refining the basic features to develop a fully matured embryo to become fully developed.
All of these developmental events happen in utero or inside the womb of the mother, and is only possible because of an amazing organ, the placenta.
The placenta is actually a temporary organ that grows during pregnancy. It’s the only dispensible organ in the human body.
The placenta attaches to the wall of the uterus and forms a connection between the mother and the developing baby. This connection is essential for providing the baby with the oxygen and the nutrients necessary for growth and for removing the waste products from the baby’s blood.
The blood vessels of the mother and the baby are found side-by-side in the placenta. This keeps their blood supplies separate. You will notice that the baby’s umbilical cord arises from the placenta.
The umbilical cord itself has two components: cord blood and cord tissue. Cord blood refers to the blood found within the umbilical cord and cord tissue refers to the umbilical cord itself.
Given the placenta is a temporary organ that develops during pregnancy, shortly after the baby is born the body also expels the placenta. In the past once the umbilical cord was cut the cord and the placenta were discarded as medical waste.
Today it is a well-known fact that cord blood, cord tissue and even the placenta are valuable sources of stem cells, and when possible should be preserved.
What are stem cells?
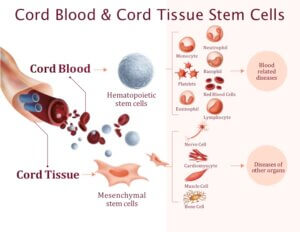 Stem cells are very early stage cells that have the capacity to grow and give rise to other types of cells in the body. They are useful for repairing damaged cells, rebuilding tissue and helping our bodies heal.
Stem cells are very early stage cells that have the capacity to grow and give rise to other types of cells in the body. They are useful for repairing damaged cells, rebuilding tissue and helping our bodies heal.
Cord blood and cord tissue are both sources of newborn stem cells. Cord blood contains hematopoietic stem cell. They give rise to other types of blood cells.
Cord tissue contains a different type of stem cell known as mesenchymal stem cells. They have the ability to give rise to bone cells, muscle cells, cartilage cells and nerve cells.
Together cord blood and cord tissue contain two different kinds of stem cells that can be used to growth a number of different cell types in the body.
Hematopoietic stem cells have been used to treat diseases for more than 60 years. The very first stem cell transplant was performed in 1957, more than 60 years ago by Dr. E. Donnall Thomas. He went on to win the Nobel Prize in 1990 for his discovery of cell transplants, as a way to treat human diseases.
Today, hematopoietic stem cells are used to treat over 80 life-threatening diseases such as cancers, immune system disorders, blood disorders and metabolic disorders.
What is hematopoietic stem cell therapy?
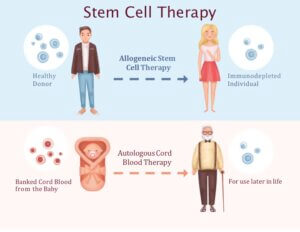 Hematopoietic stem cells therapies that are currently in use can be broadly categorized into two groups: allogeneic stem cell therapies and autologous therapies.
Hematopoietic stem cells therapies that are currently in use can be broadly categorized into two groups: allogeneic stem cell therapies and autologous therapies.
Allogeneic stem cell therapy involves collecting healthy stem cells from a donor and transplanting them into a patient as a treatment option.
Autologous stem cell therapy involves replacing the stem cell of a patient with his or her own healthy stem cells.
For example, when autologous therapy is used to treat cancer, healthy stem cells stem cells are collected and extracted from a patient prior to chemotherapy. Following chemotherapy, damaged stem cells are replaced with the patients’ own healthy stem cells.
According to a survey form2012 about 47% of hematopoietic stem cell therapies involved allogeneic stem cell transplant. This means before a patient can be treated, you have to find a matching donor to provide with a healthy stem cell sample.
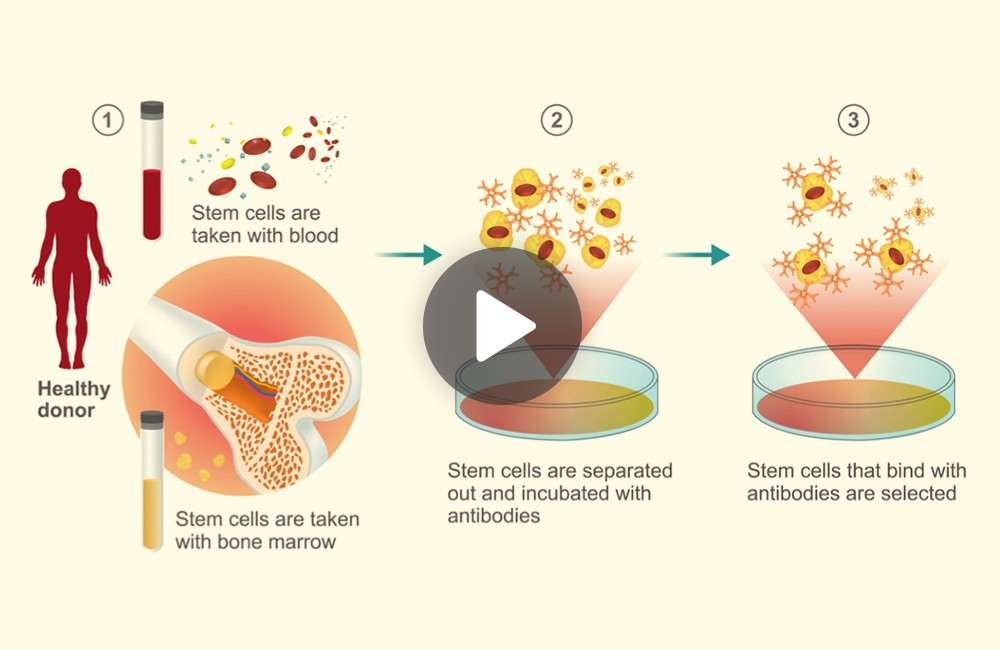
Let’s look at allogeneic stem cell therapy in more detail. The initial stages of this procedure involve taking stem cell samples from a donor either using a blood sample or in a majority of the cases a sample from the bone marrow. In the next step, stem cells from these samples are purified using specialized antibodies.
 These selected stem cells are further processed before being transplanted or infused into a patient. Next we want to highlight some of the limitations associated with allogeneic stem cell therapy
These selected stem cells are further processed before being transplanted or infused into a patient. Next we want to highlight some of the limitations associated with allogeneic stem cell therapy
First you have to find a matching donor, a step that can be extremely difficult. Even with a matched sample, there is a high risk of rejection due to what is known as the graft-versus-host disease.
This is a case where the immune system of the patient recognizes the transplanted cells as being foreign, and starts attacking them.
Next, bone marrow donation involves surgery. It is a surgical procedure, which is invasive even for the donor. The final point to consider is that the age of the donor may be relevant because even stem cells age.
Studies have compared stem cells from young versus older donors and noticed significant differences in robustness in these samples. You will want to explore our webinar on stem cells and aging if you want to learn more about this topic.
We will end this webinar by highlighting some of the potential stem cell sources that are available for cell therapies. These include adult stem cells, which can be isolated from the bone marrow and a variety of other tissues. Isolating adult stem cells can be difficult because the number of stem cells in our body decreases as we age.
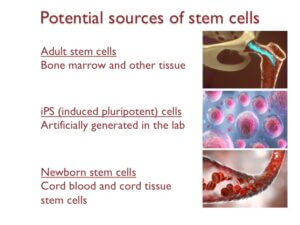 Induced pluripotent stem cells or iPS cells are another source that is being widely explored in the lab. These are stem cells made in the lab by reverse engineering adult cells.
Induced pluripotent stem cells or iPS cells are another source that is being widely explored in the lab. These are stem cells made in the lab by reverse engineering adult cells.
IPS cells have contributed to revolutionary discoveries involving stem cells in the lab, however, the safety and efficacy of using them for cell therapy remains to be established.
The final source is newborn stem cells. Newborn stem cells refer to stem cells from cord blood and cord tissue. The main limitation here is that these stem cells can only be collected at birth.
Future of stem cell therapy
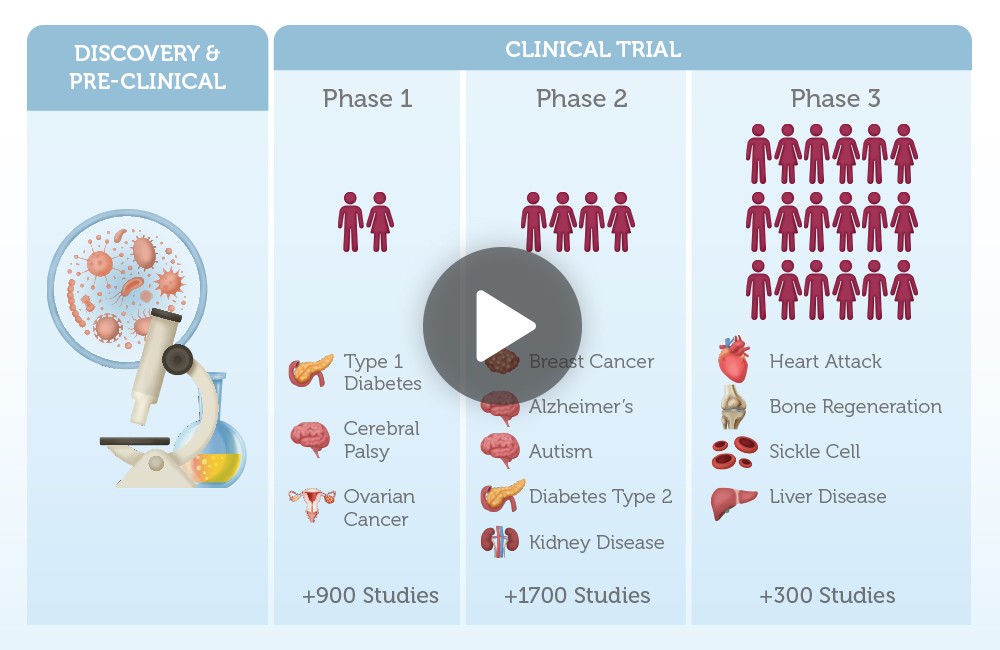
Stem cell research is one of the fastest growing branches of medical research. Currently, there are over 1000 clinical trials exploring the use of both cord blood and cord tissue stem cells to treat diseases like osteoporosis, Alzheimer’s, diabetes, breast cancer, liver failure and arthritis.
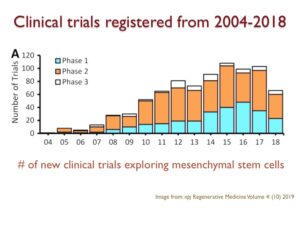 For instance, there has been a tremendous amount of growth in the number of clinical trials that were registered between 2004 to 2018. If we focus in on the clinical trials involving mesenchymal stem cells, the kind of stem cells found in cord tissue, between 2005 and 2015 the number of trials increased by 10-fold
For instance, there has been a tremendous amount of growth in the number of clinical trials that were registered between 2004 to 2018. If we focus in on the clinical trials involving mesenchymal stem cells, the kind of stem cells found in cord tissue, between 2005 and 2015 the number of trials increased by 10-fold
When we categorize these trials by stem cell types and transplant types, one thing you notices is that umbilical cord stem cells or newborn stem cells from cord blood and cord tissue, are the second largest category being explored for their potential as a future therapeutic.
It is likely that scientists are only at the tip of the iceberg in terms of developing new treatments for diseases using stem cells.
This is why the rest of the videowill focus on how clinical trials are exploring the use of stem cell therapy for treating heart disease and type 2 diabetes.
The STAR heart study
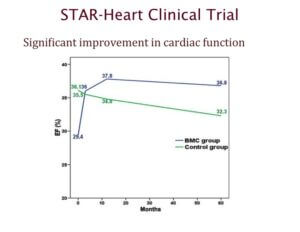 The STAR heart study is one of the largest clinical trials that looked at using stem cells to treat patients with heart disease. This trial involved 391 patients with chronic heart failure, who had experienced a heart attack 3 to 8 years prior to the study.
The STAR heart study is one of the largest clinical trials that looked at using stem cells to treat patients with heart disease. This trial involved 391 patients with chronic heart failure, who had experienced a heart attack 3 to 8 years prior to the study.
Results from this study showed that patients who received stem cells experienced significant improvement even 5 years after treatment. The graph highlighted here looks at ejection fraction, which is a measure of how much blood is pumped out by the left ventricle with each contraction. Higher numbers correspond to better efficiency.
You can see that patients who received stem cells – shown in blue – experienced an improvement in heart function after treatment, and were able to maintain these improvements in the long-term.
What’s more, there was also a significant decrease in the long-term mortality rates of patients who received stem cells after a heart attack. The average mortality rate of the group that received stem cells was 0.75% per year compared to 3.68% per year seen in the control group that did not receive stem cells.
In summary, the STAR-heart study suggests that stem cell therapy can improve heart function, quality of life, and enhance survival rates in people with chronic heart failure.
Type 2 diabetes
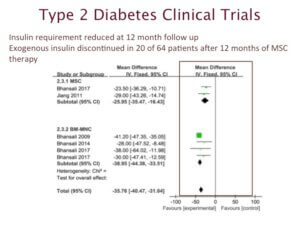 Next we want to highlight clinical trials involving type 2 diabetes and stem cell therapy. Type 2 diabetes is a metabolic disease linked to the hormone insulin. Insulin is responsible for regulating blood sugar levels.
Next we want to highlight clinical trials involving type 2 diabetes and stem cell therapy. Type 2 diabetes is a metabolic disease linked to the hormone insulin. Insulin is responsible for regulating blood sugar levels.
People with type 2 diabetes are unable to either produce insulin or their bodies have stopped responding to insulin, so their blood sugar levels remain constantly high.
The data we are highlighting, comes from 11 clinical trials involving 386 patients with type 2 diabetes. They were treated with stem cell therapy.
This is a summary of data looking at insulin requirement after 12 months of therapy. You can see that majority of the patients showed significant improvement. For example, 31% of the patients who received mesenchymal stem cells no longer required insulin injections.
Based on results from these clinical trials, we can predict that perhaps one-day stem cell therapy may become a standard of care for treating diabetes.
Now that you know more about newborn stem cells and their value we will end by highlighting the processes involved in stem cell banking. When a parent decides to preserve their baby’s stem cells we ask them to register online to receive a collection kit.
Ideally this should be done 4-6 weeks ahead of the due date. Once you register, a collection kit will be shipped right to your house. All you have to do is remember to bring it to the hospital on the day of delivery.
Once the baby is born and the umbilical cord is cut according to the birth plan of the mom, the healthcare provider can go ahead and collect the left over blood in the umbilical cord and the cord itself. Everything that is needed for these steps from the collection bags to storage vessels are included in the kit.
In the lab these samples go through many different screening steps. Once this is done stem cells are are cryogenically preserved and stored in vapour phase storage tanks. The samples will remain in storage for the lifetime of the baby or until they become necessary.
To recap, we’ve told you the amazing growth stem cell research is experiencing at the moment and how this could potentially lead to many new cell therapies in the future. We’ve also highlighted the role newborn stem cells can have in terms of advancing these studies and interms of forming form of future therapies.
We want to stress that both cord blood and cord tissue are valuable source of stem cells and they are only available at birth.
More topics you might like
Continue your journey by selecting another topic.
What are newborn stem cells and why are they important?
3:45 minutes | Stem cell basics
Understand the different types of stem cell transplants.
5:30 minutes | Stem cell basics
Learn about the exciting new ways stem cells are being explored as a future therapeutic.
5 minutes | Stem cell basics